This is an open access article, free of all copyright, and may be freely reproduced, distributed, transmitted, modified, built upon, or otherwise used by anyone for any lawful purpose. Received: MaAccepted: JPublished: August 15, 2018 This proof of concept encourages developing species-specific SynExo recombinases for genome engineering.Ĭitation: Valledor M, Myers RS, Schiller PC (2018) Herpes ICP8 protein stimulates homologous recombination in human cells. ICP8 significantly enhanced gene targeting rates in HEK 293T cells while Beta was not only unable to catalyze recombineering but inhibited gene targeting using endogenous recombination functions, despite both synaptases being well-expressed and localized to the nucleus. Here we describe stimulation of gene targeting to edit a novel fluorescent protein gene in the human genome using ICP8 and compared its efficiency to that of a “humanized” version of Beta protein from phage λ. 
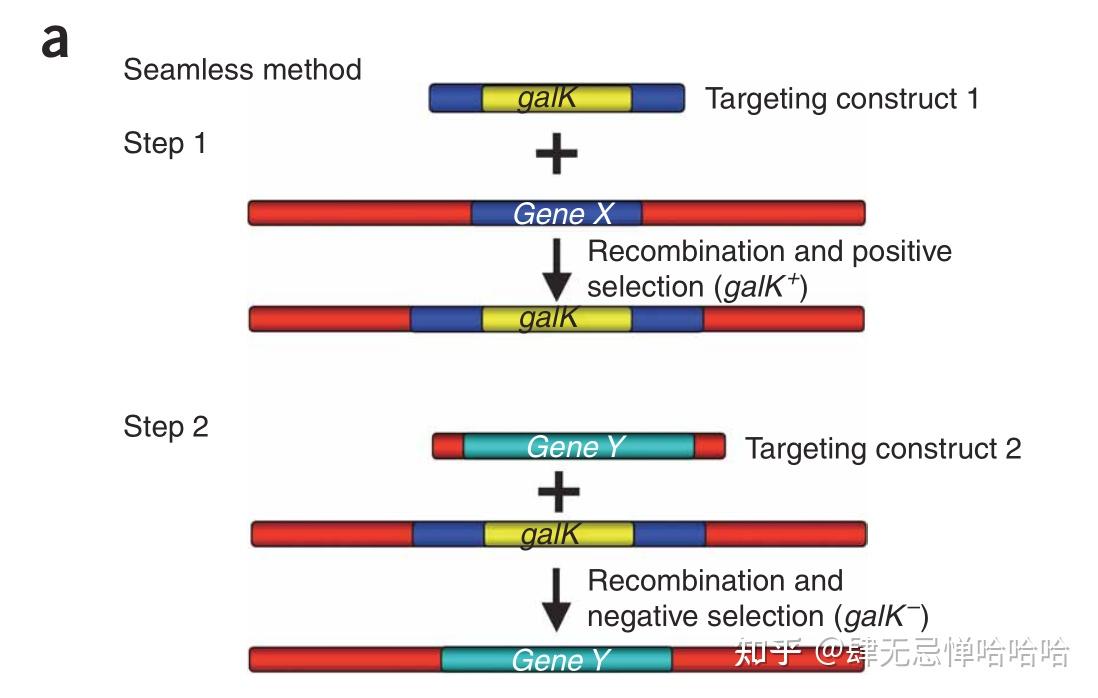
In a previous study, the Herpes SynExo was reconstituted in vitro and shown to catalyze a model recombination reaction. Human Herpes virus 1 (HHV1) encodes a SynExo comprised of ICP8 synaptase and UL12 exonuclease. SynExo recombinases are common to double-stranded DNA viruses infecting a variety of organisms, including humans. coli, recombineering using the Red Synaptase/Exonuclease pair (SynExo) in other organisms declines in efficiency roughly correlating with phylogenetic distance from E. Genome modification by recombineering using the phage lambda Red homologous recombination protein Beta in Escherichia coli has approached 100% efficiency. Recombineering has transformed functional genomic analysis.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
